What are quantum dots
Quantum dots — in Italian, quantum dots — are semiconductor nanocrystals of extremely small size, on the order of a few nanometers (millionths of a millimeter). At this minute scale, matter begins to behave differently than at larger dimensions because quantum mechanical phenomena come into play.
In practice, in these nanoparticles, normal material properties (such as color, conductivity and optical behavior) no longer depend only on the chemical composition, but on the size of the nanocrystal itself. This is why quantum dots are often described as “artificial atoms”: by confining a limited number of electrons in such a small space, quantum effects emerge that give the quantum dots unique and tunable (i.e., adjustable at will by varying their size) properties.
Indeed, one of the most fascinating features of quantum dots is their ability to emit light of different colors depending on their size. Larger quantum dots tend to emit light toward the red end of the spectrum, while smaller quantum dots emit light toward the blue or violet.
In other words, the color (i.e., wavelength) of the light re-emitted by a quantum dot can be controlled simply by changing the size of the nanocrystal. This property-known as the quantum confinement effect-allows the quantum dots to be “tuned” to the desired colors, somewhat like adjusting the notes of an instrument but in the optical domain. For example, cadmium sulfide nanoparticles of about 12 nm in diameter absorb and emit light at wavelengths typical of that material in bulk form, while reducing the particles to only ~4 nm their optical properties change markedly shifting toward blue. This sensitivity to size is why quantum dots are appreciated as “color-adjustable nanocrystals,” capable of adding a whole rainbow of possibilities to nanotechnology.
Compositionally, quantum dots typically consist of semiconductor materials. Initially, many quantum dots studied were based on cadmium compounds (such as cadmium selenide CdSe or cadmium sulfide CdS) coated by shells of other semiconductor, but to reduce their toxicity, heavy metal-free quantum dots such as those based on silicon, carbon or other elements are also being developed. The internal structure can be homogeneous (core) or core-shell (core and shell), a configuration that often improves the stability and light efficiency of the quantum dot. In any case, what matters is that each quantum dot contains only a few thousand atoms in total; to make a comparison, the size ratio between a quantum dot and a soccer is similar to that between the same football and the planet Earth. This extreme miniaturization underlies the unusual behaviors of quantum dots and their potential in a variety of fields.
How they work: quantum mechanics in a nanocrystal
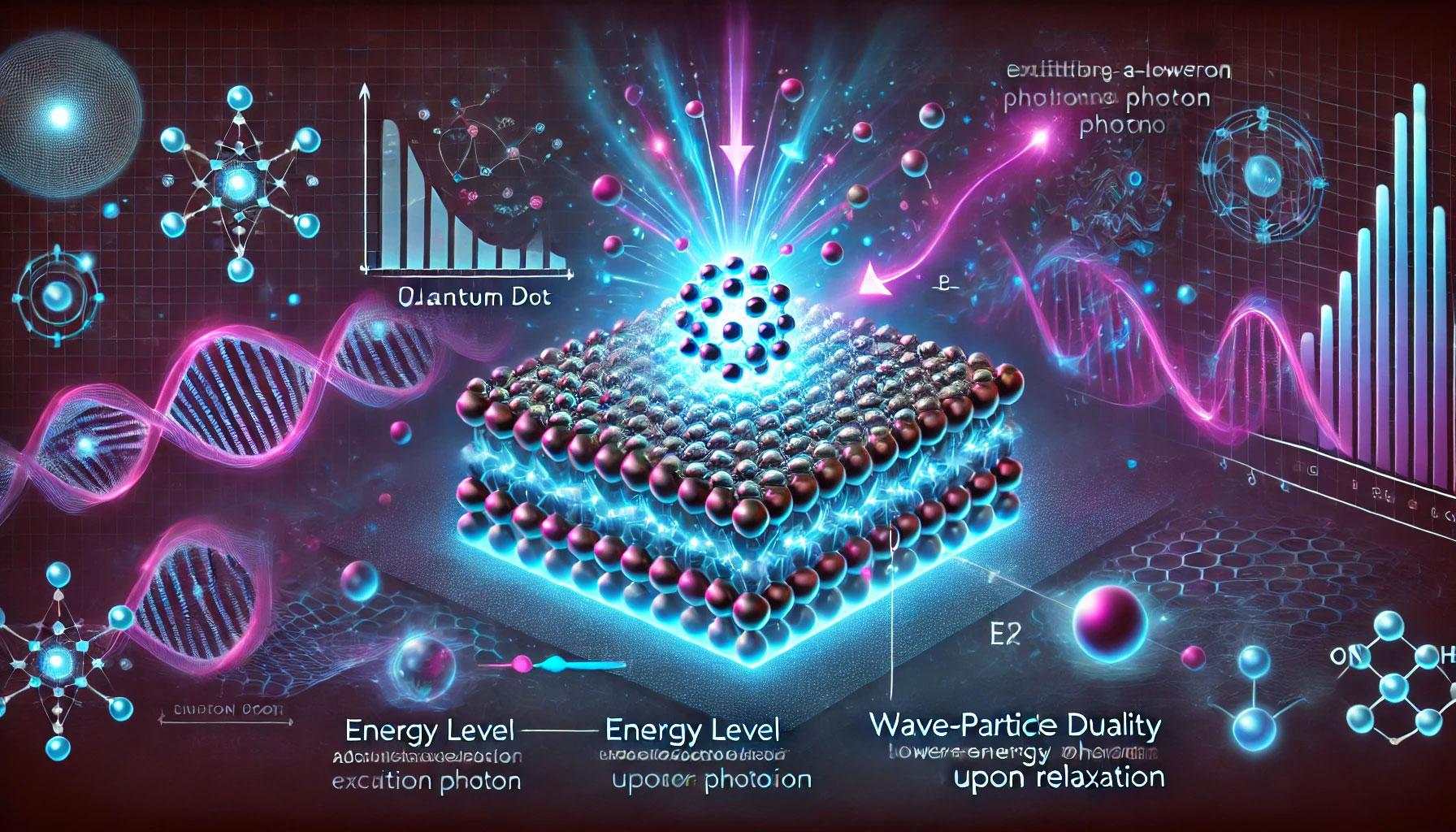
A quantum dot can absorb photons (incoming light) by exciting an electron at a higher energy level; when the electron returns to the ground state, the nanocrystal emits a photon of a certain energy (color). Since the quantized levels depend strictly on the size of the nanocrystal, it is sufficient to synthesize quantum dots of a certain size to obtain a specific emission color. This tailored emission property makes quantum dots ideal as phosphors (luminescent materials) in many optical applications. In addition, quantum dots have very broad absorption spectrum and narrow emission spectrum, which means they can be excited with a single source (e.g., UV light) to produce different pure colors. They are also significantly brighter and more resistant to photobleaching (fading under illumination) than conventional organic dyes: it is estimated that a single quantum dot can be at least ten times brighter than a conventional fluorescent molecule. These characteristics make quantum dots behave as excellent fluorophores, often superior to fluorescent dyes and proteins used in the past.
Another key aspect is the versatility of their surface. Quantum dots can be functionalized, that is, coated or conjugated with various molecules (polymers, chemical groups, etc.) to make them more stable, water-soluble, or to attach specific ligands (such as antibodies, peptides, or other recognition molecules)Another key aspect is the versatility of their surface. Quantum dots can be functionalized, that is, coated or conjugated with various molecules (polymers, chemical groups, etc.) to make them more stable, water-soluble or to attach specific ligands to them (such as antibodies, peptides or other recognition molecules). This allows quantum dots to be programmed to bind to particular targets or to be combined with other systems. For example, by coating a quantum dot with a silica shell or suitable polymers, one can make it biocompatible and equip it with functional groups (amine, carboxylic, thiol) to which biomoleculescan be attached. This chemical flexibility further broadens the possible applications, especially in the biomedical field, as we shall see in a moment.
Quantum dots in the medical and health fields
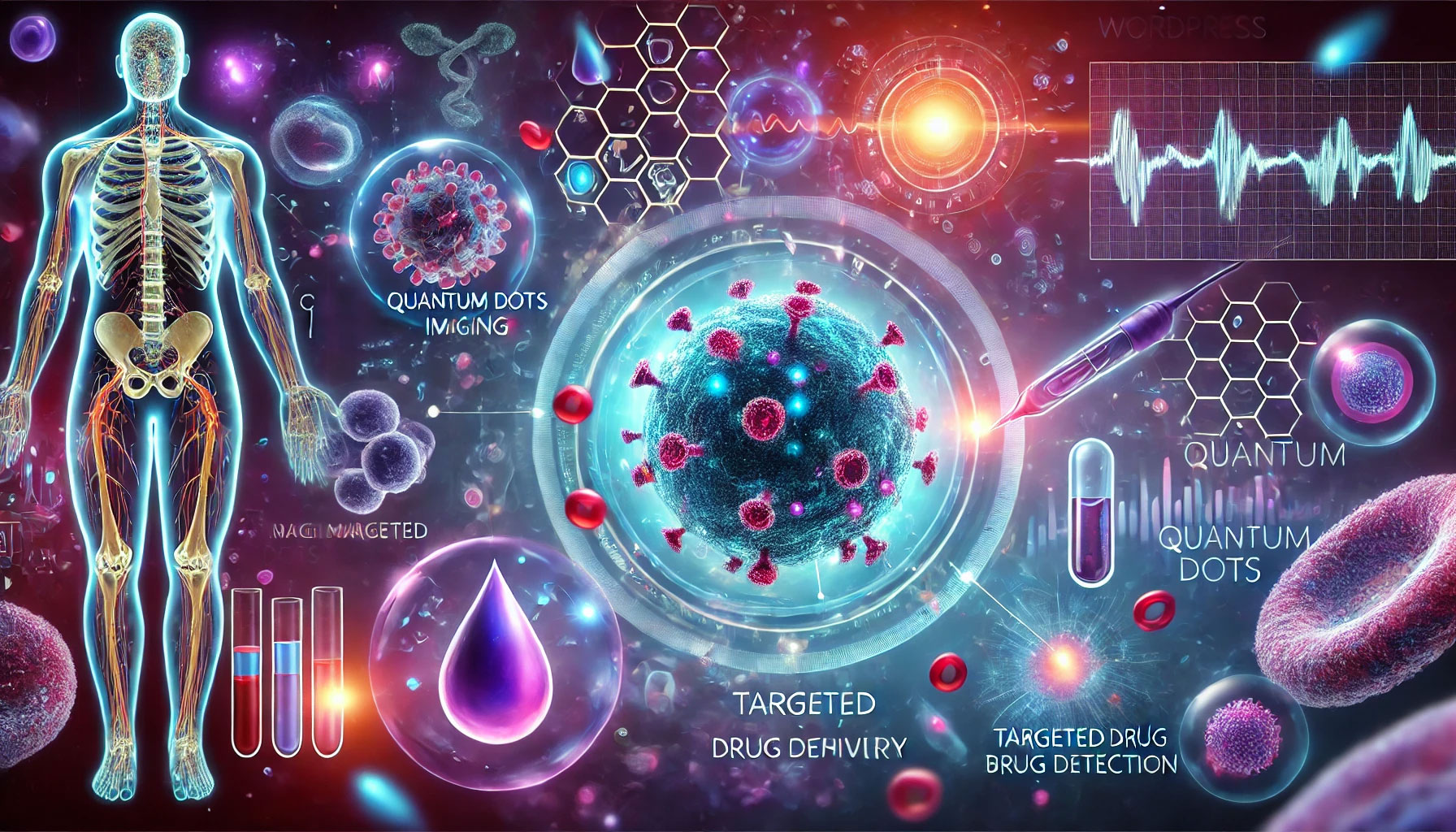
Among the most promising and studied applications of quantum dots is the medical and biomedical fields. By virtue of their special optical properties, quantum dots are revolutionizing some approaches in imaging, biological analysis, and even therapies. Here we examine how quantum dots are being used (or experimented with) in healthcare, from laboratory imaging to the prospect of new targeted therapies.
Fluorescent imaging and diagnostics: Quantum dots are used as fluorescent probes to visualize biological structures with very high sensitivity. For example, in fluorescence microscopy, QDs can be bound to specific molecules (such as antibodies directed against tumor antigens or cell markers) and introduced into tissue samples or organisms: by illuminating them with appropriate light, they will emit fluorescence allowing one to see where those target molecules are located. Because of their high brightness and stability, quantum dots make it possible to detect even very small amounts of targets and to follow their evolution over time, without fading rapidly as traditional dyes would.
In clinical-experimental settings, studies have been conducted to use quantum dots in visualizing tumors and other lesions within the body. One strategy is to conjugate QDs with antibodies that selectively recognize antigens present on tumor cells. Thus, by injecting these nanosensors into a laboratory animal, they preferentially accumulate in the tumor tissue. By then illuminating the area with near-infrared light, the tumor literally “lights up” thanks to the fluorescence of the quantum dots attached to it. For example, researchers at RIKEN in Japan have shown that quantum dots conjugated to anti-HER2 antibodies localize to breast tumors in mouse models, making the tumor mass visible as early as 24 hours after administration, while non-functionalized quantum dots do not accumulate in the tumor, or allow early diagnosis through highly sensitive fluorescent scans.
Another innovative approach is multi-modality diagnostics: combining optical quantum dot imaging with traditional radiological methods (such as PET or MRI) to obtain complementary information. For example, the use of radiolabeled quantum dots for PET has been proposed, taking advantage of their photostability and functionalizable surfaces to attach radioisotopes to them. In parallel, the same QD nanoparticle can provide a fluorescent optical signal. Combining PET and QD fluorescent imaging results in hybrid diagnostics: PET offers quantitative sensitivity on tracer accumulation in the body, while quantum dot fluorescence provides high-resolution imaging at the cellular level. In such a scenario, for example, cancer lesions could be first localized in the body with PET and then visualized in detail by fluorescent endoscopy, improving the accuracy of cancer characterization. Although these techniques are experimental, they outline the potential of QDs as versatile and powerful diagnostic tools.
Targeted therapy and theranostics: In addition to diagnosing, quantum dots can also be harnessed to treat, falling under the emerging field of “theranostics” (therapy + diagnostics). Because they can be selectively delivered to certain tissues, QDs can serve as drug shuttles or activating agents for photomediated therapies. One example is photodynamic therapy (PDT): some quantum dots can act as photosensitizers, that is, generate reactive oxygen species (ROS) when illuminated, causing localized destruction of target cells (e.g., cancer cells). Because quantum dots can be designed to absorb light at specific wavelengths, they can be calibrated to activate only where and when desired by irradiating them from outside with a laser of that frequency. This opens the way for minimally invasive anticancer therapies, in which QDs accumulated in a tumor release their toxic action only under light, limiting damage to surrounding healthy tissue.
Another line of research is the use of quantum dots as drug carriers. One can bind drug molecules to the quantum dot surface or even trap them inside a hollow quantum dot, creating pharmacoportant nanoparticles. By changing the surface chemistry, QDs can be made sensitive to certain stimuli (pH, enzymes, magnetic fields, etc.) so that they release the drug only in the presence of the desired stimulus. Imagine, for example, a quantum dot circulating in the body that is inert, but within an acidic tumor environment it is degraded by releasing a chemotherapeutic right where it is needed: this would increase the efficacy of the treatment by reducing its systemic side effects. Again, this is pre-clinical research, but the results so far indicate that quantum dots can be engineered to perform active therapeutic as well as diagnostic functions.
Finally, the aspect of biocompatibility and safety, which is crucial for any medical application, should be mentioned. Some traditional quantum dots contain elements such as cadmium or lead, which are toxic: this initially limited their use in vivo. Research is now moving toward more biocompatible quantum dots (such as carbon-based QDs, graphene quantum dots or silicon quantum dots) and toward coatings that seal out toxic components, preventing their release into the body. Preliminary studies suggest that, with proper design precautions, QDs can be used safely at least diagnostically. For example, carbon-based quantum dots (carbon dots) have shown low toxicity and are already used in some research-level imaging tests. Of course, further testing and licensing will be needed before widespread clinical use, but the high interest in biomedical applications of quantum dots is accelerating these developments.